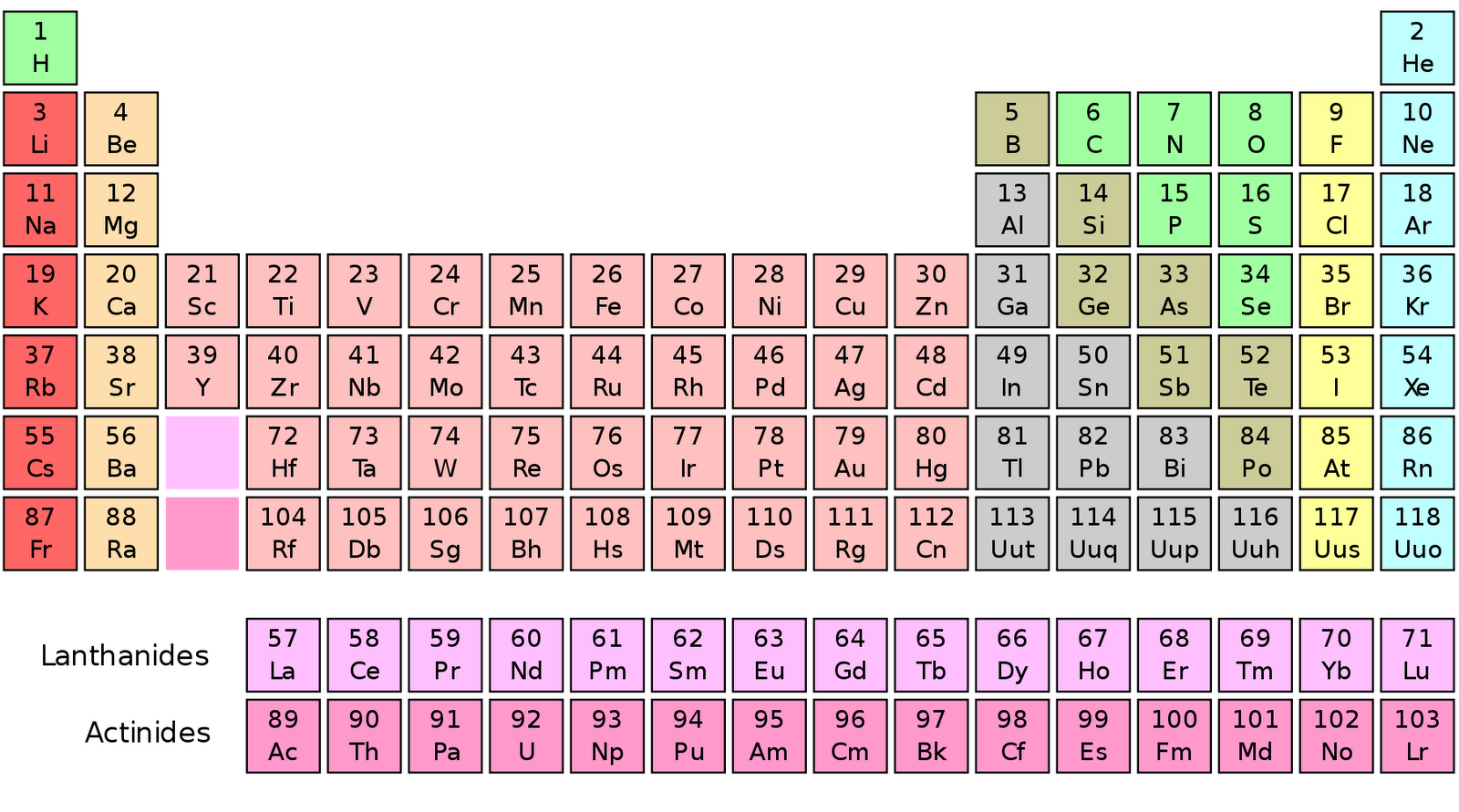 
You will need to refer to a periodic table for proton values. This is a periodic table with atomic mass, element name, element symbol, and atomic number. In this notation, the atomic number is not included. You can see that in ZnCl2, there is 1 Zinc atom and 2 Chlorine atoms. 2 Now, to calculate the molar mass of ZnCl2, you just have to add the molar mass of all the individual atoms that are present in ZnCl2. Interactive periodic table with up-to-date element property data collected from authoritative sources. For example, the elements in AgCl are silver and chlorine. For O: (16 x 3) 48 4th step: Now, the molar mass would be: 54 g/mol + 48 g/mol 102 g/mol Summary: To find the molar mass in periodic table, we do four steps.First of all, we identify the elements in the compound. Symbol-mass format for the above atom would be written as Cr-52. The molar mass of Chlorine is 35.45 g/mol. (A r of the element x number of atoms of the element). Periodic table-basic Created Date: 11:50:10 AM. Parenthesis indicate mass of most stable nuclide 1 2. For an example of this notation, look to the chromium atom shown below:Īnother way to refer to a specific atom is to write the mass number of the atom after the name, separated by a hyphen. Atomic masses obtained from the National Institute of Standards and Technology atomic masses with more than 5 decimal places have been rounded. The "A" value is written as a superscript while the "Z" value is written as a subscript. Both the atomic number and mass are written to the left of the chemical symbol. The composition of any atom can be illustrated with a shorthand notation called A/Z format. Our PTE App is the ultimate tool for every friend of chemistry - whether pupil or teacher, student or professor, amateur or expert, hobbyist or technician. Where #M# is the molar mass in grams per mole ( #gmol^-1#), #m# is the mass of the sample in grams ( #g#), and #n# is the amount in moles of the sample ( #mol#).\nonumber \] Immediately find any imaginable information about an element, calculate molar masses of any compounds you like, facilitate your overview with infographics and much more. Note that some masses are in parentheses, usually towards the bottom of the table of elements, The parentheses basically mean that scientists don’t. Generally, this number is the one with the decimal point. In the periodic table, the vertical columns are called groups and the horizontal rows are called periods. The table has mass, or molar mass, shown as the number in the bottom of each square. Periodic Table of Elements - The periodic table is the tabular arrangement of all the chemical elements on the basis of their respective atomic numbers. Helium, with two protons, will have two electrons. 
Hydrogen atoms all have one electron occupying the space outside of the nucleus. Since atoms are neutral, the number of electrons in an atom is equal to the number of protons. Ask your teacher or tutor if you are not sure which one to use.Īnother way to find molar mass if amount and mass of a substance are known is by using the formula Periodic Table with Mass Numbers and Atomic Symbols. 1: The social security number subatomic-the proton. The calculator utilizes the periodic table of elements to determine the atomic weights of the elements present in the formula. To use the calculator, simply input the chemical formula of the substance and click the 'Calculate' button. 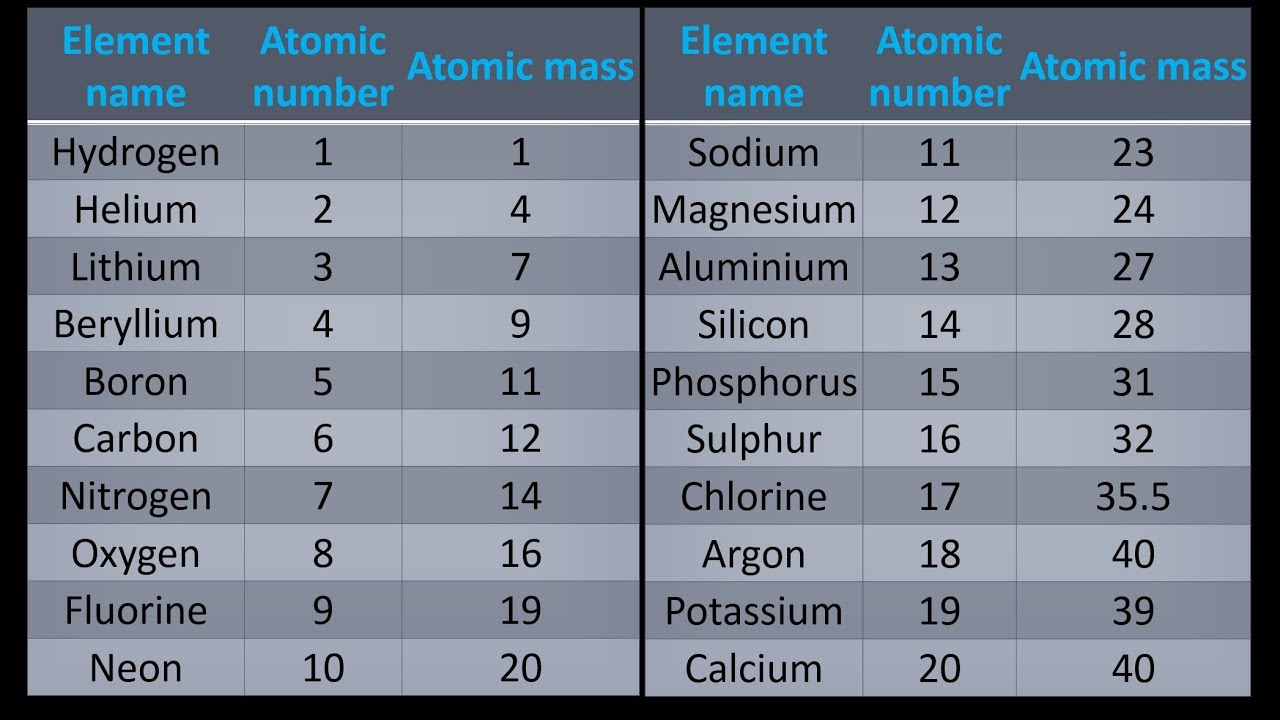
The molar masses will be slightly different if you are using a table with more accurate decimal values for atomic mass. Molar mass is the mass of one mole of a substance and is expressed in grams per mole. Sulfuric acid #H_"2"SO_4# has 2 Hydrogen atoms (mass #1#), 1 sulfur atom (mass #32#) and 4 oxygen atoms (mass #16#) so: You can see that The molar mass of Sodium is 22.990 g/mol. So let’s look at the molar mass of Sodium and Oxygen from the above periodic table. Now in Na2O, there are 2 Sodium atoms and 1 Oxygen atom. Then simply find out how many of each atom is in the molecule and add the respective atomic masses to find the molecular or molar mass.įor example water #H_2O# has 2 Hydrogen atoms (mass #1#) and 1 oxygen atom (mass #16#) so: You can see the molar mass value of all the atoms from this periodic table. I prefer to use tables with whole number atomic masses, but that is just personal preference. Find a periodic table and look at the atomic mass of each atom in the molecule. Download the latest PDF version of the periodic table with standard atomic weights and isotopic abundances, updated by IUPAC.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
